Definition
A carbohydrate (pronounced /kɑ:bəˈhaɪdreɪt/) is an organic compound with the empirical formula Cm(H2O)n; that is, consists only of carbon, hydrogen, and oxygen, with a hydrogen:oxygen atom ratio of 2:1 (as in water). Carbohydrates can be viewed as hydrates of carbon, hence their name. Structurally however, it is more accurate to view them as polyhydroxy aldehydes and ketones.
The term is most common in biochemistry, where it is a synonym of saccharide. The carbohydrates (saccharides) are divided into four chemical groupings: monosaccharides, disaccharides, oligosaccharides, and polysaccharides. In general, the monosaccharides and disaccharides, which are smaller (lower molecular weight) carbohydrates, are commonly referred to as sugars.[1] The word saccharide comes from the Greek word σάκχαρον (sákkharon), meaning "sugar". While the scientific nomenclature of carbohydrates is complex, the names of the monosaccharides and disaccharides very often end in the suffix -ose. For example, blood sugar is the monosaccharide glucose, table sugar is the disaccharide sucrose, and milk sugar is the disaccharide lactose (see illustration).
Carbohydrates perform numerous roles in living things. Polysaccharides serve for the storage of energy (e.g., starch and glycogen), and as structural components (e.g., cellulose in plants and chitin in arthropods). The 5-carbon monosaccharide ribose is an important component of coenzymes (e.g., ATP, FAD, and NAD) and the backbone of the genetic molecule known as RNA. The related deoxyribose is a component of DNA. Saccharides and their derivatives include many other important biomolecules that play key roles in the immune system, fertilization, preventing pathogenesis, blood clotting, and development.[2]
In food science and in many informal contexts, the term carbohydrate often means any food that is particularly rich in the complex carbohydrate starch (such as cereals, bread, and pasta) or simple carbohydrates, such as sugar (found in candy, jams, and desserts).
Structure
Formerly the name "carbohydrate" was used in chemistry for any compound with the formula Cm(H2O)n. Following this definition, some chemists considered formaldehyde CH2O to be the simplest carbohydrate,[3] while others claimed that title for glycolaldehyde.[4] Today the term is generally understood in the biochemistry sense, which excludes compounds with only one or two carbons.
Natural saccharides are generally built of simple carbohydrates called monosaccharides with general formula (CH2O)n where n is three or more. A typical monosaccharide has the structure H-(CHOH)x(C=O)-(CHOH)y-H, that is, an aldehyde or ketone with many hydroxyl groups added, usually one on each carbon atom that is not part of the aldehyde or ketone functional group. Examples of monosaccharides are glucose, fructose, and glyceraldehyde. However, some biological substances commonly called "monosaccharides" do not conform to this formula (e.g., uronic acids and deoxy-sugars such as fucose), and there are many chemicals that do conform to this formula but are not considered to be monosaccharides (e.g., formaldehyde CH2O and inositol (CH2O)6).[5]
The open-chain form of a monosaccharide often coexists with a closed ring form where the aldehyde/ketone carbonyl group carbon (C=O) and hydroxyl group (-OH) react forming a hemiacetal with a new C-O-C bridge.
Monosaccharides can be linked together into what are called polysaccharides (or oligosaccharides) in a large variety of ways. Many carbohydrates contain one or more modified monosaccharide units that have had one or more groups replaced or removed. For example, deoxyribose, a component of DNA, is a modified version of ribose; chitin is composed of repeating units of N-acetylglucosamine, a nitrogen-containing form of glucose.
Monosaccharides
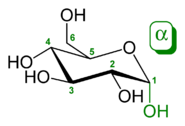
Monosaccharides are the simplest carbohydrates in that they cannot be hydrolyzed to smaller carbohydrates. They are aldehydes or ketones with two or more hydroxyl groups. The general chemical formula of an unmodified monosaccharide is (C•H2O)n, literally a "carbon hydrate." Monosaccharides are important fuel molecules as well as building blocks for nucleic acids. The smallest monosaccharides, for which n = 3, are dihydroxyacetone and D- and L-glyceraldehyde.Classification of monosaccharides
Monosaccharides are classified according to three different characteristics: the placement of its carbonyl group, the number of carbon atoms it contains, and its chiral handedness. If the carbonyl group is an aldehyde, the monosaccharide is an aldose; if the carbonyl group is a ketone, the monosaccharide is a ketose. Monosaccharides with three carbon atoms are called trioses, those with four are called tetroses, five are called pentoses, six are hexoses, and so on.[6] These two systems of classification are often combined. For example, glucose is an aldohexose (a six-carbon aldehyde), ribose is an aldopentose (a five-carbon aldehyde), and fructose is a ketohexose (a six-carbon ketone).
Each carbon atom bearing a hydroxyl group (-OH), with the exception of the first and last carbons, are asymmetric, making them stereocenters with two possible configurations each (R or S). Because of this asymmetry, a number of isomers may exist for any given monosaccharide formula. The aldohexose D-glucose, for example, has the formula (C·H2O)6, of which all but two of its six carbons atoms are stereogenic, making D-glucose one of 24 = 16 possible stereoisomers. In the case of glyceraldehyde, an aldotriose, there is one pair of possible stereoisomers, which are enantiomers and epimers. 1,3-dihydroxyacetone, the ketose corresponding to the aldose glyceraldehyde, is a symmetric molecule with no stereocenters). The assignment of D or L is made according to the orientation of the asymmetric carbon furthest from the carbonyl group: in a standard Fischer projection if the hydroxyl group is on the right the molecule is a D sugar, otherwise it is an L sugar. The "D-" and "L-" prefixes should not be confused with "d-" or "l-", which indicate the direction that the sugar rotates plane polarized light. This usage of "d-" and "l-" is no longer followed in carbohydrate chemistry.
Disaccharides

(Sucrose, also known as table sugar, is a common disaccharide. It is composed of two monosaccharides: D-glucose (left) and D-fructose (right).)
Two joined monosaccharides are called a disaccharide and these are the simplest polysaccharides. Examples include sucrose and lactose. They are composed of two monosaccharide units bound together by a covalent bond known as a glycosidic linkage formed via a dehydration reaction, resulting in the loss of a hydrogen atom from one monosaccharide and a hydroxyl group from the other. The formula of unmodified disaccharides is C12H22O11. Although there are numerous kinds of disaccharides, a handful of disaccharides are particularly notable .
Sucrose, pictured to the right, is the most abundant disaccharide, and the main form in which carbohydrates are transported in plants. It is composed of one D-glucose molecule and one D-fructose molecule. The systematic name for sucrose, O-α-D-glucopyranosyl-(1→2)-D-fructofuranoside, indicates four things:
- Its monosaccharides: glucose and fructose
- Their ring types: glucose is a pyranose, and fructose is a furanose
- How they are linked together: the oxygen on carbon number 1 (C1) of α-D-glucose is linked to the C2 of D-fructose.
- The -oside suffix indicates that the anomeric carbon of both monosaccharides participates in the glycosidic bond.
Lactose, a disaccharide composed of one D-galactose molecule and one D-glucose molecule, occurs naturally in mammalian milk. The systematic name for lactose is O-β-D-galactopyranosyl-(1→4)-D-glucopyranose. Other notable disaccharides include maltose (two D-glucoses linked α-1,4) and cellulobiose (two D-glucoses linked β-1,4). disaccharides can be classified into two types.They are reducing and non-reducing disaccahrides if the functional group is present in bonding with another sugar unit it is called as reducing disaccharide.
Oligosaccharides and polysaccharides

( Amylose is a linear polymer of glucose mainly linked with α(1→4) bonds. It can be made of several thousands of glucose units. It is one of the two components of starch, the other being amylopectin.)
Oligosaccharides and polysaccharides are composed of longer chains of monosaccharide units bound together by glycosidic bonds. The distinction between the two is based upon the number of monosaccharide units present in the chain. Oligosaccharides typically contain between three and ten monosaccharide units, and polysaccharides contain greater than ten monosaccharide units. Definitions of how large a carbohydrate must be to fall into each category vary according to personal opinion. Examples of oligosaccharides include the disaccharides mentioned above, the trisaccharide raffinose and the tetrasaccharide stachyose.
Oligosaccharides are found as a common form of protein posttranslational modification. Such posttranslational modifications include the Lewis and ABO oligosaccharides responsible for blood group classifications and so of tissue incompatibilities, the alpha-Gal epitope responsible for hyperacute rejection in xenotransplantation, and O-GlcNAc modifications.
Polysaccharides represent an important class of biological polymers. Their function in living organisms is usually either structure- or storage-related. Starch (a polymer of glucose) is used as a storage polysaccharide in plants, being found in the form of both amylose and the branched amylopectin. In animals, the structurally similar glucose polymer is the more densely branched glycogen, sometimes called 'animal starch'. Glycogen's properties allow it to be metabolized more quickly, which suits the active lives of moving animals.
Cellulose and chitin are examples of structural polysaccharides. Cellulose is used in the cell walls of plants and other organisms, and is claimed to be the most abundant organic molecule on earth.[9] It has many uses such as a significant role in the paper and textile industries, and is used as a feedstock for the production of rayon (via the viscose process), cellulose acetate, celluloid, and nitrocellulose. Chitin has a similar structure, but has nitrogen-containing side branches, increasing its strength. It is found in arthropod exoskeletons and in the cell walls of some fungi. It also has multiple uses, including surgical threads.
Other polysaccharides include callose or laminarin, chrysolaminarin, xylan, arabinoxylan, mannan, fucoidan and galactomannan.














0 comments:
Post a Comment